OVERVIEW
Cough monitoring
Cough monitoring has been moving away from subjective patient self-reporting in favor of more objective and quantitative measures. As regulators continue to look for more granular cough and lung sound metrics, we continue to partner with device manufacturers to bring sponsors the most advanced respiratory trial solutions to meet these needs.
For which indications is cough typically assessed?
Cough and lung sounds are evaluated for a number of respiratory diseases:
- Asthma
- Chronic Obstructive Pulmonary Disease (COPD)
- Chronic bronchitis
- Cardiopulmonary diseases
- Allergies
- Gastroesophageal Reflux Disease (GERD)
- Sleep disorders and nocturnal wheezing
- Postnasal drip
SOLUTION FOR
Objective cough and lung sound monitoring
Clario supports accurate, in-home monitoring of cough and lung sounds through RESP® Biosensor, a lightweight, wearable device able to capture 24 hours of continuous cough recordings. Used in combination with the Clario iSpiro® home spirometry solution, the RESP Biosensor enables sponsors to gain objective cough metrics alongside spirometry and eCOA data.
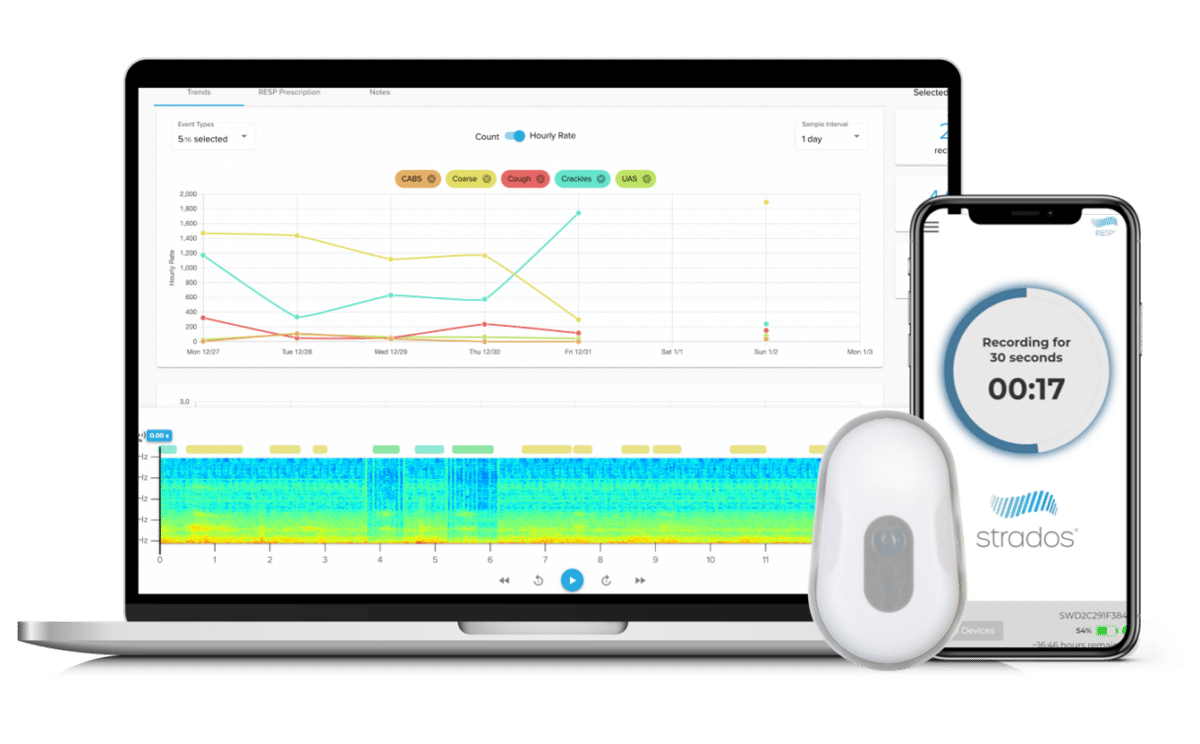
RESP® Biosensor System
Allows for the following endpoints:
- Cough count, severity and spasms
- Lung sounds: wheezing, crackles, rhonchi, snore and others
- On-board accelerometer allows for concurrent measurements*:
- Respiratory rate and heart rate
- Activity levels
- Body positioning
Benefits of the Clario and Strados Labs Solution:
- iSpiro, RESP and eCOA applications all on the same handheld device
- Continuous recording of up to 25 hours on a single charge
- Small, comfortable, hands-free device with no wires
- Clinically validated, FDA 510(k) Cleared, CE Marked Device
- Validated overread process supported by machine learning algorithms*
- HIPAA-compliant, encrypted data storage and transfer

Patient-friendly wireless device
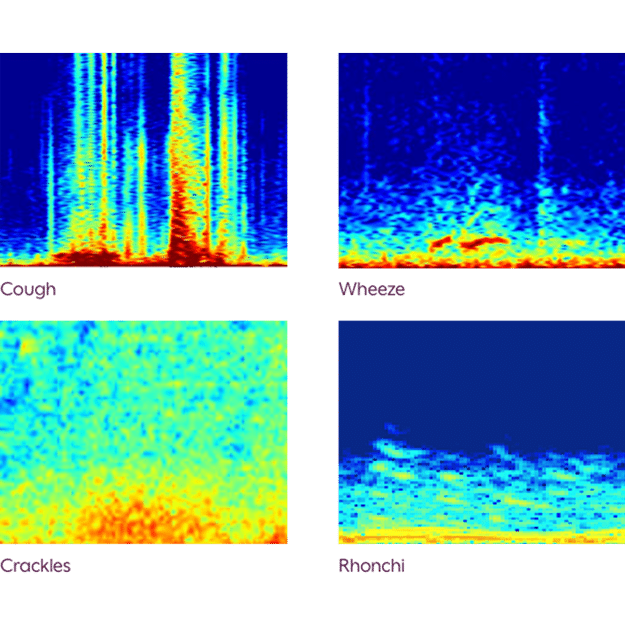
Lung sound events are displayed visually as spectrograms to facilitate the overread process*
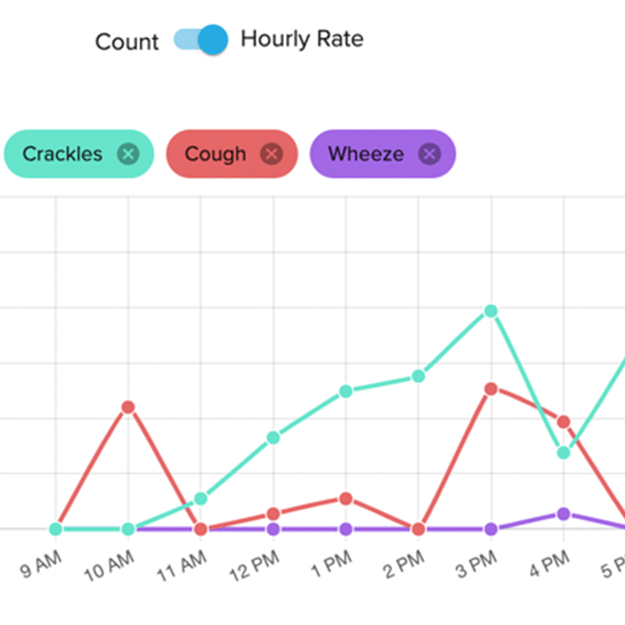
Granular data over a 24-hour period with concurrent measurement of various respiratory endpoints
*Algorithms, respiratory rate and chest excursions not yet FDA cleared
RESP Biosensor metrics at-a-glance
1st
510(k)-cleared wearable device for lung sounds
24h
continuous recording
19M
breaths captured
600k
validated coughs
OVERVIEW VIDEO
Learn more about the benefits of Strados Labs’ RESP Biosensor System in clinical trials
Related solutions
Peer-reviewed journals
Artificial intelligence outperforms pulmonologists in the interpretation of pulmonary function tests
Artificial Intelligence Assists in Quality Assessment of Spirometry in Clinical Trials
Comparing performance of primary care clinicians in the interpretation of SPIROmetry with or without Artificial Intelligence Decision support software(SPIRO-AID): a protocol for a randomized controlled trial
FAQs
Why are cough and lung sound endpoints used in clinical trials?
Objective measures such as cough frequency, cough intensity, and lung sound analysis are increasingly utilized as clinically meaningful endpoints in respiratory research. These parameters provide non-invasive, quantifiable markers of airway pathology and disease activity, offering a level of precision that complements traditional pulmonary function tests and patient-reported outcomes. Objective cough measures are complementary to patient reported data as they are more accurate and eliminate challenges of patient recall.
Their utility is particularly evident in conditions characterized by significant symptom burden, including refractory chronic cough, chronic obstructive pulmonary disease (COPD), asthma, and interstitial lung diseases. In these populations, subjective assessments alone may fail to fully capture treatment effects or subtle changes in disease trajectory.
By incorporating acoustic and frequency-based analyses, these endpoints enable continuous, real-world monitoring of respiratory symptoms, thereby enhancing the sensitivity of clinical trials to detect therapeutic benefit and improving the overall understanding of disease progression.
How are cough and lung sound data collected?
Data acquisition for cough frequency and lung sound analysis is facilitated through the use of a wearable monitoring device, such as the Strados RESP® Biosensor.
This advanced sensor system is designed to continuously capture acoustic signals associated with respiratory activity, including cough events and characteristic lung sounds, in real-world settings. The collected data undergoes algorithmic processing using validated analytical models that quantify cough frequency and intensity, as well as classify lung sounds into clinically relevant categories (e.g., wheeze, crackles).
This approach provides objective, high-resolution endpoints that enhance the sensitivity of clinical trials and support comprehensive evaluation of disease burden and therapeutic response. By leveraging continuous monitoring and automated analysis, the RESP® Biosensor enables accurate, reproducible, and non-invasive assessment of respiratory symptoms, thereby reducing reliance on subjective reporting and improving overall data quality.
Talk to a specialist to get started
Our team of experts is available to address questions you may have about our Respiratory solutions. Submit your contact information and we’ll be in touch shortly.