Cardiac safety concerns are among the leading reasons that promising drugs are delayed or abandoned. We offer a range of options tailored to your compound, site locations and ever-changing regulatory requirements.
Product detail
Patch Arrhythmia
- Leveraging our deep Holter arrhythmia knowledge combined with more patient-friendly devices, Clario provides global logistics and clinical trial quality data. We have flexible options for recording durations up to 14 days and offer wire-free single and dual channel recording options.
Holter Arrhythmia
- Continuous 12 lead ECG Holter solution to produce detailed data for 24-48hr increments
- Over 40 years of experience in Holter arrhythmia analysis enables Clario to provide the most consistent, reliable arrhythmia data
Related solutions

Blood Pressure Services
Collect and centrally analyze accurate, consistent, quality blood pressure data from site-based to decentralized trials.

ECG — On-site to DCT and Phase I-IV
Monitor every heartbeat with precision from anywhere to assess safety and efficacy throughout your site-based or decentralized clinical trial.
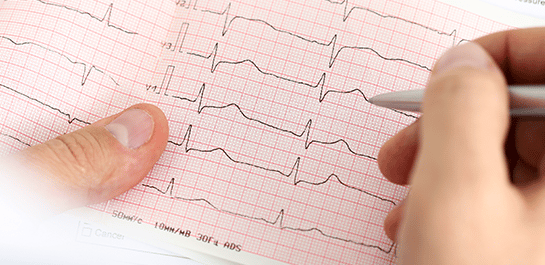
Phase I/TQT Cardiac Assessment
Fulfill your ICH E14 requirement early, minimize risk and potentially reduce costs throughout your trial by collaborating with our Phase 1 Center of Excellence.

Regulatory Consultation and Statistical Analysis Expertise
Leverage our industry key opinion leaders to understand the complex regulatory landscape while minimizing risk with confidence.
Learn more or speak with one our experts
Our team of clinical trial cardiac safety experts is always available to address any questions you may have about our cardiac safety solutions. Submit your contact information and we’ll be in touch shortly.
