Clario Survey Reveals Challenges and Innovations within CNS Clinical Trials
Results demonstrate adoption of virtual and wearable technologies to increase effectiveness of CNS studies
PHILADELPHIA – February 25, 2021 – Clario, the global leader in clinical endpoint data collection, today released a new report, Innovation Frontiers in CNS Research, which analyzes the results of a recent industry survey on central nervous system (CNS) clinical trials. The report reveals key challenges for sponsors and study teams, as well as the innovations they are implementing to address them, including wearable technologies and virtual trial solutions.
CNS diseases include all manner of neurological and psychiatric disorders and conditions and represent one of the largest economic, social and capital burdens worldwide of any disease group. In fact, one in five U.S. adults (51.5 million) and one in six U.S. adolescents (7.7 million) are living with some type of CNS condition.1,2 The prevalence of CNS disorders indicates a significant need for continued therapeutic innovation. However, unique challenges specific to the therapeutic area, such as the many unknowns about the diseases themselves, have challenged progress in the search for effective treatments and cures.
Fielded in the fourth quarter of 2020, the survey seeks to better understand how a cross-section of key industry professionals from leading pharmaceutical companies are adopting and using new innovations within CNS trials to address challenges specific to this therapeutic area. Key findings include:
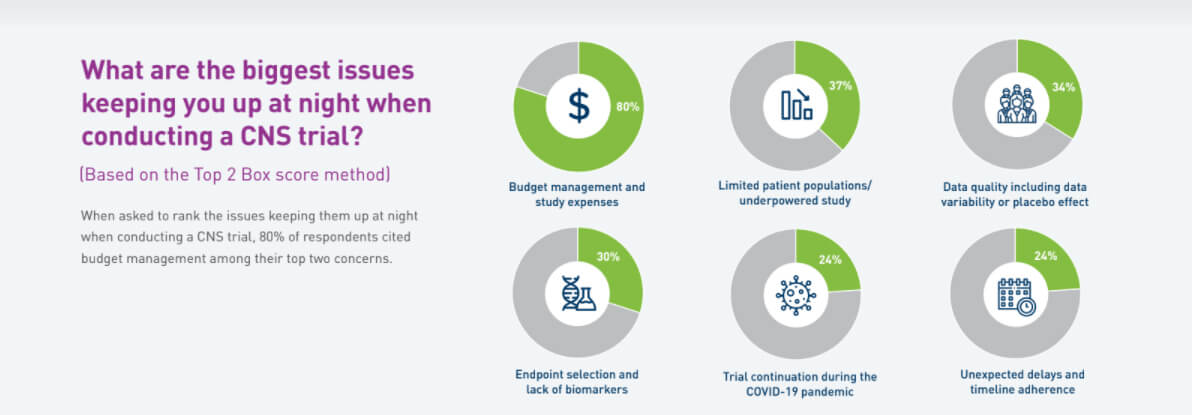
What keeps you up at night?: When asked to rank the issues keeping them up at night when conducting a CNS trial, 80% of respondents cited budget management among their top two concerns.
Current or planned use of wearables: 80% of survey respondents have used, or are planning to use, wearable technology in their CNS Trials.
Current adoption of virtual trial technologies in CNS trials: More than one third of respondents report running fully virtual, or decentralized, CNS trials or a hybrid model featuring some virtual solutions.
“CNS therapies represent a pressing societal need. Manufacturers are looking to innovative patient centric technologies that also offer patients improved convenience like telehealth and at-home data collection to accelerate and improve trial execution” said Jim Mahon, Executive Vice President, Chief Product, Strategy and Marketing Officer, Clario.
To learn more, download the report, Innovation Frontiers in CNS Research, which includes detailed survey findings and strategies for operationalizing and scaling trial innovations.
About Clario
Clario delivers the leading endpoint technology solutions for clinical trials. Through experience gained from over 19,000 clinical trials delivered in support of 870 regulatory approvals, Clario fuses scientific expertise and global scale into the broadest endpoint technology platform to enable pharmaceutical, biotech and medical device partners to transform lives. Clario has mastered the ability to generate rich evidence across all trial models: decentralized, hybrid and site-based clinical trials. With 30 facilities in nine countries across North America, Europe and Asia Pacific, Clario’s global team of science, technology and operational experts has been delivering the richest clinical evidence for nearly 50 years.
For more information, go to Clario.com or follow us on LinkedIn and Twitter.
Clario Media Contact
1 National Institute of Mental Health
2 “US National and State-Level Prevalence of Mental Health Disorders and Disparities of Mental Health Care Use in Children,” JAMA Pediatrics, Feb. 11, 2019.