Simplify complexity with a single-partner solution
No matter the phase, region or level of complexity, Clario can support your oncology trial with scientific, operational, and technology expertise.




Experience speed and simplicity with our solutions
Focus on your compound while we expertly manage and simplify your endpoint data strategy. By partnering with us you can:
- Demonstrate efficacy with high-quality data
- Understand complex regulatory requirements
- Meet critical milestones
- Capture safety data efficiently and accurately
- Minimize site and patient burden

Get end-to-end support
Biotech sponsors rely on us to support them throughout their oncology drug development lifecycle. We provide:
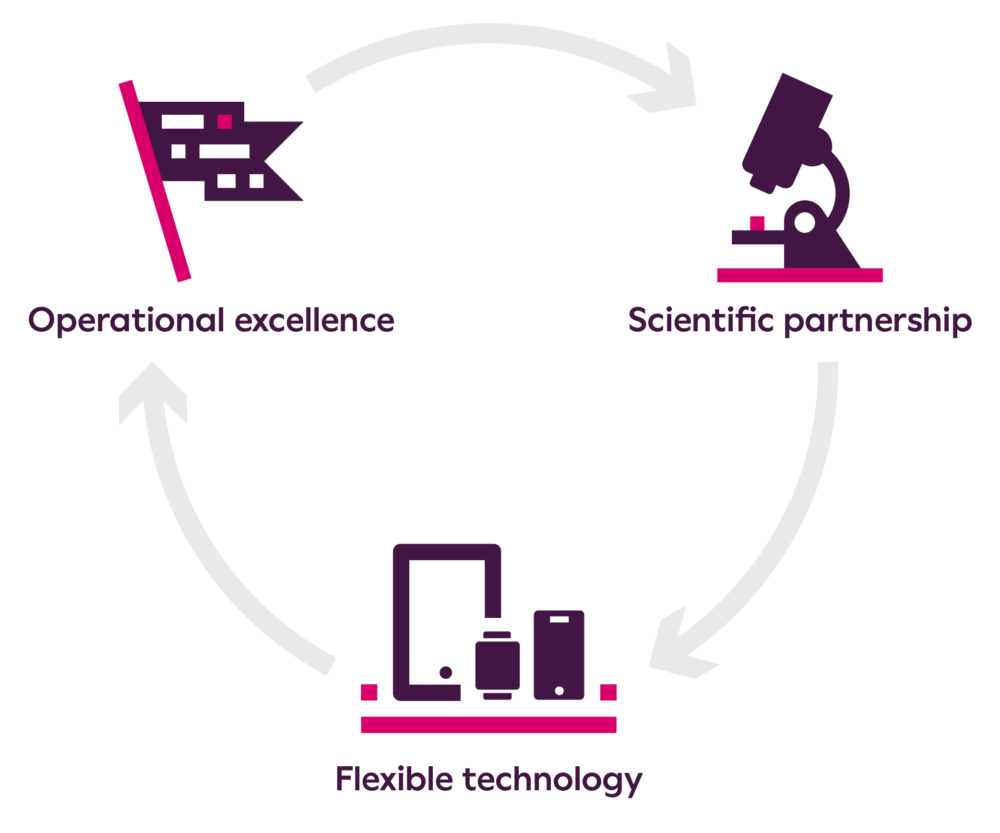
- Scientific partnership: Giving guidance on endpoint data strategy to optimize efficacy, safety, and quality-of-life measurements while identifying areas of differentiation.
- Flexible technology: Across cardiac safety, eCOA/ePRO, imaging, and wearables for hybrid, remote, or in-clinic trials.
- Operational excellence: Project Managers across regional time zones, expert logistics capabilities, and real-time access to your study data.
Read a real-life example of our end-to-end support.
SUPPORTED
OVER
75%
of FDA new drug approvals for oncology trials since 2012
Delivering results for biotechs
Our experience and expertise mean we understand the unique pressures biotech companies like yours face — and how to solve for them.
Experience across early phase and pivotal trials for submission
Operation across every major geographic region
Delivery of high-quality endpoint data across imaging, eCOA, cardiac safety, and more
Ready to learn how to simplify complexity and move faster toward your next milestone?
Unlock the full potential of your oncology trial with Clario by your side.