FREE ON-DEMAND WEBINAR
Stop letting outdated data capture compromise your oncology trial
In early phase oncology trials, every data point matters. Smaller patient populations, compressed timelines and high-stakes investment demand early, accurate signal detection. But manual, outdated data capture slows progress and creates preventable risk.
Watch the free replay of our webinar to learn how modern endpoint data capture methodologies and technologies can help you protect data integrity.


In this exclusive webinar,
learn how modern endpoint methodologies and technologies can help you:
- Detect early efficacy signals sooner
- Uncover safety risks more accurately
- Optimize dosing decisions with greater confidence
- Differentiate your compound with stronger evidence
All while reducing trial delays, hidden costs, and uncertainty.
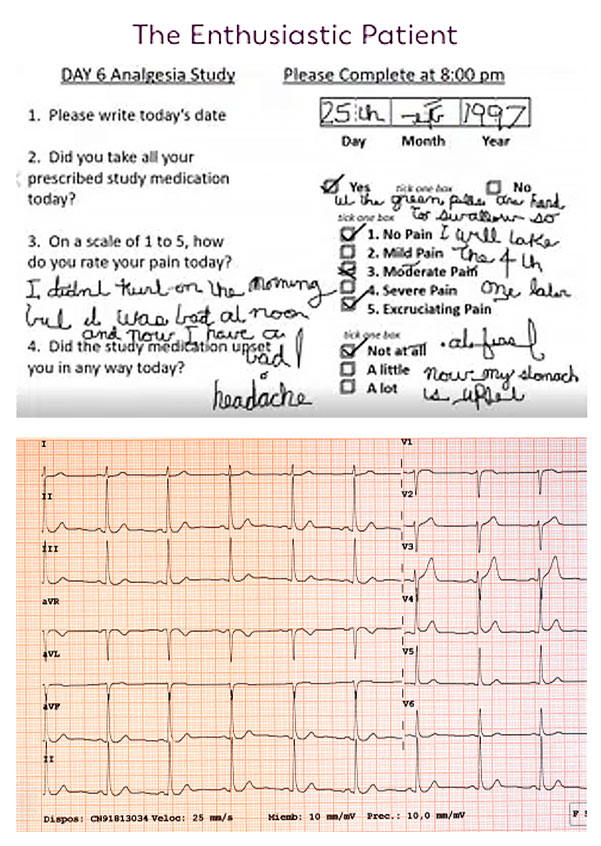
How antiquated data capture holds back trials
Traditional data capture methods create risk:
- Slower, variable cardiac safety measurements:
Paper, site-based ECGs can increase false inclusions and miss the detection of potential cardiotoxicity issues. - Weaker efficacy evidence:
Site-based imaging may be inconsistent, resulting in early signal detection being overlooked and making dosing decisions harder. - Delayed, poor-quality patient impact data:
Paper Patient-Reported Outcomes (PROs) delay the detection of symptomatic adverse events and may miss quality-of-life data.
Result: Reduced confidence, slower go/no-go decisions, and increased trial uncertainty.
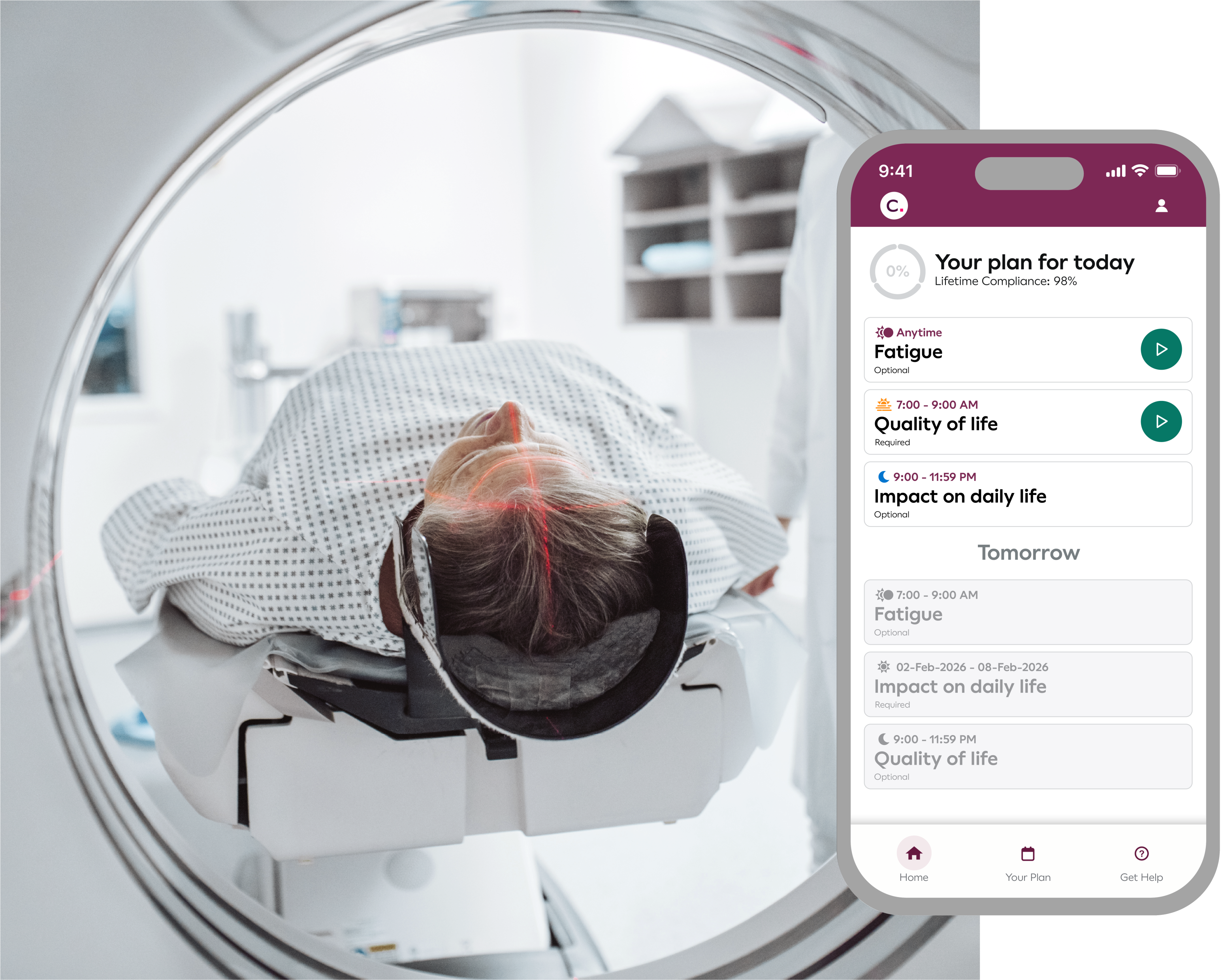

Reduce risk with earlier, more accurate insights
Clario’s advanced imaging, cardiac safety, and eCOA solutions deliver earlier, more actionable insights:
Identify safety signals more reliably:
Cardiac safety evaluation and electronic PROs reveal risks earlier and with greater precision, protecting patients.
Detect early efficacy signals more accurately:
Advanced imaging delivers more sensitive response evaluation and tumor burden assessments.
Optimize dosing selection and understanding of tolerability levels:
Pinpoint optimal anti-tumor activity for evidence-driven dose decisions, while capturing patient tolerability.
Differentiate your drug and meet regulatory expectations:
Capture your drug’s impact on patients’ quality of life accurately and reliably with ePROs.
Move beyond traditional data capture
Discover how to maximize the value of your early phase oncology data.